Magnetism in Gemstones
An Effective Tool and Method for Gem Identification
Color Change Garnets
"Champagne" "Peach" Brown Gold
Green Purple Blue Pink
Green Color Change Garnets are the only green transparent gems besides Demantoid Garnets that pick up with an N52 magnet. The high magnetic susceptibility in green Color Change Garnets is due primarily to manganese (Mn2+), as opposed to iron (Fe3+) in Demantoids.
Although it has not been described in other gemological literature, some Color Change Garnets can show up to four different colors under visible light (as opposed to UV light) depending on the type of visible light applied. An example is the Color Change Garnet pear pictured below. This gem changes from purple (daylight) to gray (fluorescent light) to green (white LED light) to pink (incandescent light).
Blue Color Change Garnet
in Daylight (Madagascar)
Blue Color Change Garnet
in Incandescent Light (Madagascar)
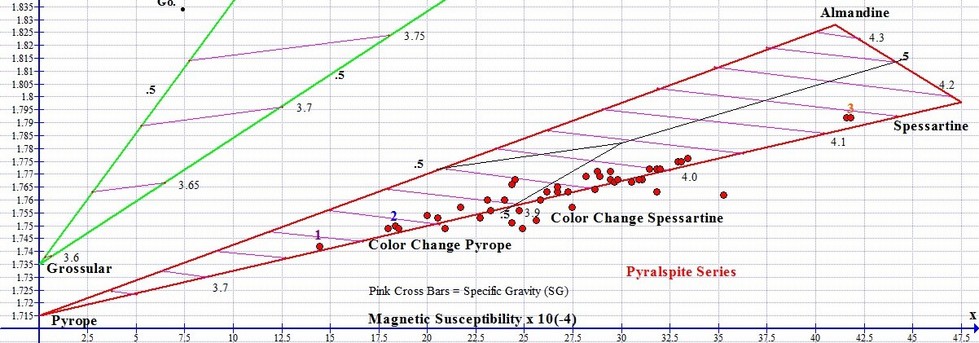
Results of our Garnet study suggest that most Color Change Garnets with orange daylight color fall in the Spessartine trisection, and we classify them as Color Change Spessartines. An extreme example of a Color Change Garnet that is primarily composed of Spessartine is the brownish orange trillion pictured below. This gem is represented on the graph above by the far-right graph point labeled #3. The major composition of this Color Change Garnet overlaps that of standard orange Spessartine gems, but unlike typical Spessartine, the color changes to red under incandescent light.
We classify this extreme example as a Color Change Garnet, but it could also be classified as a rare example of Spessartine Garnet that is chromium-bearing. The presence of chromium enables the color change.
Chromium can be detected in this gem as red fluorescence under blue laser light (405nm), and chromium is also clearly detected with a spectrometer. Fluorescence in Garnets is usually restricted to the two allochromatic species Pyrope and Grossular, and as far as we know, fluorescence in idiochromatic Spessartine has never been reported. A similar example of chromium-bearing Spessartine from Madagascar that showed color change was reported in 2002 (Schmetzer, et al. 2002), but no fluorescence in that gem was reported.
Green Color Change Garnet
Orange Color Change Garnet in Daylight
Color Change Pyropes
The seven Color Change Pyropes shown in the above photo are ordered from left to right to show increasing magnetic susceptibility due to increasing Spessartine content. There is no predictable graduation of color, because most colors are controlled by low concentrations of chromium/vanadium rather than progressively higher concentrations of Spessartine (manganese). The measured refractive index range for Color Change Pyropes is R.I 1.742-1.768, and the magnetic susceptibility range is SI 14.45- 24.53. Broader R.I. and SI ranges are seen for Color Change Spessartine gems, which have ranges of RI 1.751-1.792 and SI 24.35- 41.79.
© Kirk Feral 2011, All Rights Reserved. These materials may be duplicated for educational purposes only. No part of this website may be duplicated or distributed for profit, for commercial purposes, or for posting to another website without the expressed written consent of the copyright holder.
Graph of Color Change Garnets (Pyrope & Spessartine)
Blue Color Change Garnet: Blue is a rare and fairly-recently discovered color in Garnet. A striking color change from daylight to incandescent light is seen in the .86 ct blue Color Change Garnet from Madagascar pictured below, which changes from blue to pink. This contrast between colors is just as impressive as the color change seen in the finest Alexandrite Chrysoberyl. Below right, we see that the blue body color is emphasized when viewed under cool LED light.
This Purple Color Change Garnet Shows 4 Colors Under 4 Different Light Sources
Not all Garnet varieties along the Pyrope-Spessartine join are Color Change Garnets. Other hybrids of Pyrope and Spessartine that may not show color change include Pastel Pyrope at the low end, Malaya in the middle, and orange Spessartine at the high end. But Color Change Garnets show the most diverse range of daylight color and incandescent color. Varying amounts of manganese (Spessartine) mixing with small amounts of iron, chromium, and vanadium, result in a rainbow of colors including blue, green, purple, brown, yellow and orange. With the exceptions of blue, green and some purple gems, color tone tends to be light or pale. The 3 Color Change Garnet gems pictured below are shown in daylight.
In our study of Garnets, we have found examples of the phenomenon of color change within every gem Garnet species except Uvarovite. In addition to the Garnets that are considered Color Change by gemologists, color change can also occur among Almandine, Spessartine, Andradite, Grossular, and Pyrope (Malaya and Pastel Pyrope varieties). However, the variety name “Color Change Garnet” is generally reserved for Pyrope-Spessartine Garnets, which at times show the most definitive color change. With compositions that fall along the join between Pyrope and Spessartine in the Pyralspite ternary, we see a nearly continuous isomorphic replacement between Pyrope and Spessartine. This researcher has tested Color Change Garnets with compositions ranging from a low of 30% Spessartine to a high of 89% Spessartine.
© Kirk Feral
Color Change Garnet
Color Change Garnet is an unusual variety of the Pyralspite series primarily found in African countries such as Madagascar, Kenya and Tanzania, but also in Sri Lanka and Afghanistan. This Garnet variety is found in every color except red and black. The body color of Color Change Garnets is modified toward the red end of the spectrum when incandescent light is applied. This is because incandescent light itself casts a “warm” reddish hue that brings out the red color related to minor amounts of chromium within the Color Change Garnets. Also due to the presence of chromium, most Color Change Garnets, regardless of body color, appear red or reddish under a Chelsea filter.
Purple Color Change Garnet
Brown Color Change Garnet
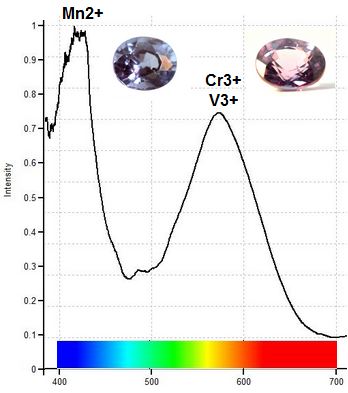
The spectrometer graph below illustrates how chromium/vanadium in the presence of manganese creates the blue daylight color and red incandescent color in blue Color Change Pyrope from Madagascar. Absorbance of light in daylight is seen below as the graph peaks for manganese (Mn2+) and chromium/vanadium (Cr3+/V3+). Blue light is transmitted from the valley or transmission window near 475nm, and transmission of red light occurs in the transmission window near 700nm. The area of transmission of blue light in the visible portion of the light spectrum is a bit greater than the area of transmission of red light, translating to predominantly blue color in daylight. But when reddish incandescent light is applied, the window of red light transmission shifts to the left, increasing the amount of red light transmitted so that pink becomes the dominant gem color.
Chromium/Vanadium in Relation to Manganese
Creates Blue and Red Colors
In our study, blue Color Change Garnets from Tanzania such as the 0.74ct gem pictured below show less of a complete change in color than do blue gems from Madagascar. The Tanzanian gems change from blue in daylight to purple rather than to pink in incandescent light.
Blue Color Change Garnet from Tanzania
Daylight and Incandescent Light
Blue Color Change Garnet
in LED Light (Madagascar)
Although green Garnet is typically associated with the Ugrandites rather than Pyralspites, green Garnet is occasionally seen as a variation of Color Change Garnet (shown below). The green Garnet trillion below fluoresces bright red under blue laser light (405 nm) due to significant chromium content.
Daylight & 405nm Fluorescence
Green Color Change Garnet
Daylight & Fluorescence
Color Change Spessartine, 1.02ct
Bekily, Madagascar
Most Color Change Garnet gems with blue or purple color fall in the Pyrope trisection, and these can be classified as Color Change Pyropes. The lowest point on the graph above (#1) is a purple Color Change Garnet pear (pictured below left) that is 69% Pyrope. The blue oval Color Change Garnet from Madagascar pictured below (right) is 63% Pyrope, as represented by point #2 on the graph above. Both are Color Change Pyropes.
Blue Color Change Pyrope, .87ct
Bekliy, Madagascar
Purple Color Change Pyrope, 1.04ct
Madagascar
Two Different Species: The graph below shows compositions for 51 Color Change Garnets of various colors tested in our study. Some Color Change Garnets, such as the blue Garnet from Madagascar shown above, have plot points that fall to the left of the half-way mark along the Pyrope-Spessartine join, and therefore have a composition that is primarily Pyrope. These gems can be classified as Color Change Pyrope, a variety of Pyrope.
More often among Color Change Garnets, graph points fall to the right of the half-way point toward Spessartine, as does the blue Garnet from Tanzania shown above, indicating that Spessartine is the primary component. These gems can be classified as Color Change Spessartine, a variety of the Spessartine species. Some Color Change Spessartines are nearly pure orange in daylight. GIA currently makes no species distinction between Color Change Garnets that are primarily Pyrope and those that are primarily Spessartine, and instead classifies most Color Change Garnets as members of an intermediate species they call Pyrope-Spessartine.
There are no red Garnets that are considered Color Change Garnets, although some red Garnets may appear a brighter shade of red under incandescent light. Color Change Garnets with cool body colors in daylight (purple, blue, green) show the most dramatic color changes, altering toward warmer colors such as red, pink or purple when incandescent light is applied. Gems with body colors that are already on the "warm" end of the spectrum (brown, yellow, orange, pinkish orange) show changes that are more subtle, becoming reddish orange or reddish pink in incandescent light. Such changes can at times be difficult to see. The orange Color Change Garnet pictured below looks similar to Spessartine Garnet in daylight and changes color slightly to orangey pink in incandescent light.
Fluorescence: Most published gemological resources incorrectly describe all Garnets other than Grossulars as non-fluorescing. In fact, many Garnets, including most Color Change Garnets, show pink to red fluorescence under blue laser light (405nm). Some Malaya Garnets and Pastel Pyropes also show fluorescence.
Fluorescence in these Pyralspite garnets is related to the presence of chromium, and is permitted by a relatively low concentration of iron. In many cases, the presence of chromium can be confirmed by a red Chelsea filter reaction. In other cases, no Chelsea filter reaction can be seen (as in some brown and orange Color Change Spessartine Garnets). In these cases, only trace amounts of chromium are present, and such low chromium concentrations contribute nothing to daylight gem color and nothing to magnetic susceptibility. The author suspects that longwave UV fluorescence in many low-iron Color Change Garnets may be quenched by vanadium rather than by iron.