"Verdelite" Tourmaline 4.1ct.
"Rubellite" Tourmaline 8.1ct.
When testing Garnets, we can accurately determine the major composition of individual gems by graphing refractive index and magnetic measurements on solid solution ternary diagrams much like those we show for Tourmalines (for a Garnet diagram, see page 1 of Garnets). For a variety of reasons, this method of determining Garnet composition is not well-suited to Tourmaline species. But we might expect that the compositions of most gem Tourmalines would fall somewhere within the Elbaite-Schorl-Dravite ternary or the Elbaite-Schorl-Tsilaisite ternary. At the present time, the best we can do is to show the measured magnetic susceptibility of each individual Elbaite gem on a bar graph.
Tourmaline is a diverse mineral group composed of 32 species currently known to mineralogists (Henry, D.J. et al., 2011). Small variations in chemical composition account for the multitude of species. Most gem-quality Tourmalines belong to only 2 species, Elbaite and to a lesser extent Dravite. Both species are allochromatic and transparent. Color varieties and trade names of Elbaite gems include green "Verdelite", red "Rubellite", blue "Indicolite", pink Tourmaline and yellow "Canary" Tourmaline. Brown gems often belong to the Dravite species, as well as green Chrome Tourmalines. Black color in Tourmaline gems is typical of Schorl, an opaque idiochromatic species.
The metal ions that cause color in any particular Tourmaline gem can only be determined by advanced lab tests such as microprobe analysis or laser ablation. However, specific colors have been found by researchers to be indicative of specific metal ions and intervalence charge transfer processes.
Pink Tourmaline 3.5ct.
"Indicolite" Tourmaline 5.3ct.
© Kirk Feral 2013, All Rights Reserved. These materials may be duplicated for educational purposes only. No part of this website may be duplicated or distributed for profit, for commercial purposes, or for posting to another website without the expressed written consent of the copyright holder.
The bar graph below shows the magnetic susceptibility (degree of magnetic attraction) of different Tourmaline color varieties in relation to one another. Each column of dots represents a color variety, presented from least magnetic to most magnetic. Dot colors are representative of gem colors, and each dot represents an individual gem's measured magnetism in terms of volume magnetic susceptibility noted as SI (International System of Units) X 10(-6). The SI values range from less than zero (< 0 SI, Diamagnetic response) to SI 547 (Drag response) for transparent Tourmaline gems, and from SI 773 (Drag response) to SI 990 (Drag response) for opaque Tourmaline gems. Gems with mixed colors are not represented on this graph.
Relative Magnetic Susceptibilities of Tourmalines by Color
All Tourmalines tested for this study had refractive indices in the range of 1.61-1.645, with the exception of iron-rich Schorl, which had a higher refractive index range of 1.63-1.67. Elbaite chemically mixes with other Tourmaline species such as Schorl, Tsilaiste, Liddicoatite and Rossmanite in what are referred to as solid solution series (see table below). Calcium-rich gems that are primarily Liddicoatite can appear identical to Elbaite gems. Dravite is in solid solution with Uvite, and many Dravite gems are a mixture of Uvite and Dravite.
Some of these species are idiochromatic, such as black Schorl (iron, Fe2+), yellow Tsilaisite (manganese, Mn2+), green Chromium-Dravite (chromium, Cr3+) and green Vanadium-Dravite (Vanadium, V3+). These are idiochromatic species that have transition metals as part of their chemical formulas. As gem species, Elbaite and Dravite are allochromatic, deriving most of their color and paramagnetism from the idiochromatic species with which they blend.
YellowTourmaline 2.5ct.
This is not the first table created to show magnetic susceptibilities of Tourmalines. In 1944, researcher A. Sigamony published a paper titled The Magnetic Properties of Tourmaline and Epidote in which he included a table of magnetic susceptibilities for 11 Tourmalines based on color. The values published by Sigamony were obtained by measuring loss of weight using an ALNICO magnet and a Curie balance, which produces measurements that are significantly less precise than what we obtain with a neodymium magnet and Hoover balance used for the current study. Also, in the classic 1985 monograph on Tourmalines titled The Tourmaline Group, R. Dietrich published a table of magnetic susceptibilities for 38 Tourmalines based on color. That table is a compilation of data from mineralogists who published from the 1940’s through the 1970’s. Those first limited efforts undertaken decades ago by mineralogists who relied on less sensitive instruments to measure gem magnetic susceptibilities are remarkably consistent with the results of our current study.
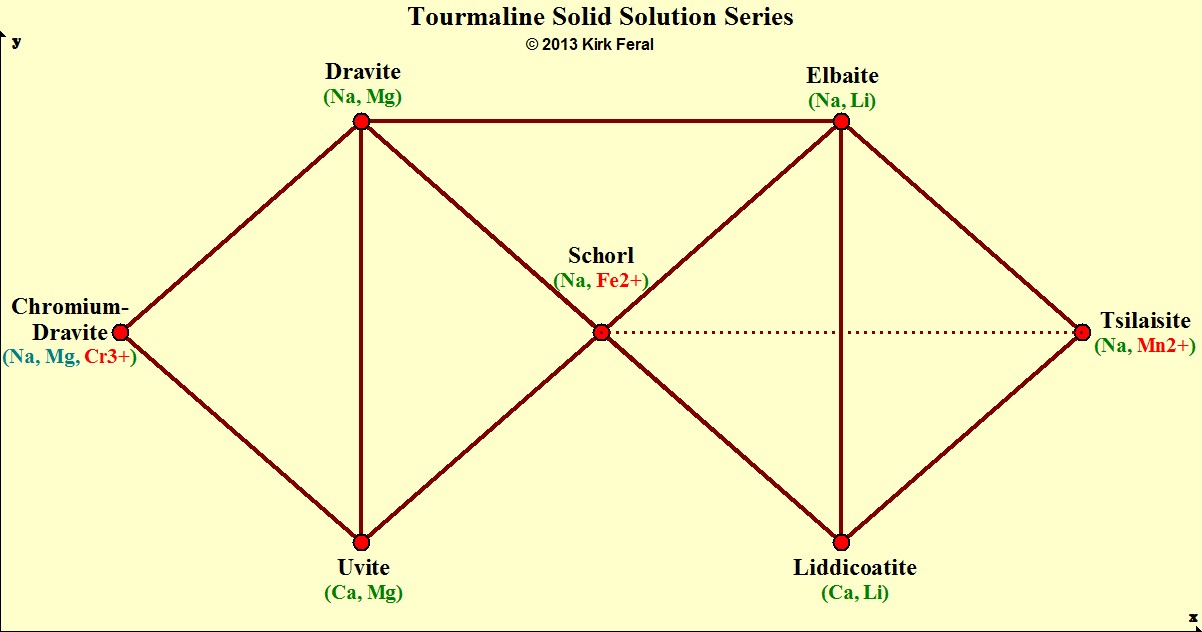
Tourmaline Species Mix Together in Solid Solution Series.
The Chromophores for 3 Idiochromatic Species are Highlighted in Red.
Contents of this Section
P.1) Overview: Tourmaline SpeciesP.2) Black, Blue and Green Tourmalines
P.3) Pink, Red, Yellow and Other TourmalinesP.4) Paraiba Tourmalines
Tourmalines:
Color and Magnetism
Overview: Tourmaline Species
Because different Tourmaline species have overlapping refractive indices, densities and magnetic susceptibilities, they cannot be definitively separated from one another using standard gemological tools. More advanced lab analysis such as Raman spectroscopy is needed to separate Tourmalines by species based on their chemical compositions. For this reason, our study of Tourmalines separates gems primarily by color rather than by species. Our assumption is that most faceted gem-quality Tourmalines are Elbaite, most Chrome Tourmalines and most brown/orange gems are Dravite, and most opaque black gems are Schorl.
The table of ternaries (triangles) below illustrates some of the many possible solid solution series among gem Tourmalines. The dotted line represents the Schorl-Tsilaisite solid solution. Vanadium-Dravite and Rossmanite species are not shown on this table.
"Chrome" Tourmaline 5.4ct
Dravite.
Bi-color Tourmaline 2.2ct
Elbaite
"Rubellite" Tourmaline 7.5ct.
Elbaite
Tourmaline gems have complex chemistry compared to Diamond, Sapphire, Ruby and Spinel, and they often contain high concentrations of iron and/or manganese. Tourmalines are the most magnetic of all natural transparent gemstones that are allochromatic (colored by impurities). In contrast, idiochromatic Garnets are far more magnetic. For our study of magnetism, we examined over 165 Tourmaline gems in a wide range of colors and shades. The findings presented here include magnetic responses to a magnet as well as magnetic susceptibility measurements taken for each Tourmaline using a Hoover balance.
1 >
Uvite: The Uvite Tourmaline species often contains inclusions and is rarely faceted as a gemstone. Uvite intermixes with the Dravite species, and the two species cannot be individually identified or separated without advanced testing. According to the sellers of the two red Brazilian Tourmalines shown below, these gems are from deposits known to be Uvite. Both gems are strongly magnetic due to iron (Fe2+) derived from mixing with Schorl.
Brownish Red Uvite (1.47ct, Strong, SI 243)
Red Uvite (0.46ct, Strong, SI 218)
Tourmaline species mix with each other in the same way that Garnet species do. Even though Elbaite Tourmaline is considered to be allochromatic, it might be more instructive to describe this gem species as partly allochromatic and partly idiochromatic, as are Grossular Garnet, Pyrope Garnet and even Peridot (green Forsterite). As the diagram above shows, Elbaite gems (with the exception of copper-bearing Elbaite) technically do not derive color and magnetic attraction from impurities as do most allochromatic gems. Elbaite gems derive color and paramagnetism by mixing with idiochromatic Tourmaline species that contain transition metals in their native chemistry. The blue color in the Elbaite Tourmaline below ("Indicolite" variety) is not caused by impurities of iron, but instead by iron derived from mixing with Schorl Tourmaline.
Blue "Indicolite" Tourmaline is primarily composed of Allochromatic Elbaite Tourmaline mixed with a lesser amount of Idiochromatic Schorl Tourmaline
Information presented here about causes of Tourmaline color was resourced from Dr. George Rossman's CalTech web page on Tourmaline spectroscopy, as well as our own analyses using a spectrometer. The causes of color in Tourmalines can be complex and subtle and remain only partially understood by researchers.
Uvite Tourmaline also mixes with the Vanadium-Dravite and Chromium-Dravite species. According to the seller of the highly included green Tourmaline rough crystal shown below, this specimen is green Uvite from Burma.
Green Uvite Crystal
6.53ct, Diamagnetic
Using spectroscopy, fluorescence, Chelsea filter testing and magnetic testing, it appears that the coloring agent in the above green Tourmaline crystal is primarily vanadium, with some chromium. This Uvite above does not fluoresce under longwave UV light, but it does fluoresce weak yellow under shortwave light. This Uvite crystal is diamagnetic.
Fluorescence
Fluorescence is rare among Tourmalines. We have found only a few examples that show fluorescence under longwave or shortwave UV light. Although colored mostly by vanadium, some green Chrome Tourmalines of the Dravite species can fluoresce weak to moderate red under longwave UV light due to the chromium component. On occasion, light blue and light green copper-bearing (Paraiba) Tourmalines of the Elbaite species can fluoresce bluish white under longwave UV light due to an unidentified activator. Under shortwave UV light, some brown Dravite, green Vanadium Dravite, and green Vanadium Uvite Tourmalines are known to fluoresce a weak yellow color. The brown Dravite below from Afghanistan is diamagnetic, and fluoresces yellow under shortwave UV light due to an undetermined activator.
Brown Dravite, 2.11ct.
Daylight & Shortwave UV Light
(Inert, SI <0)
Although Tourmaline has only modest brilliance as a gemstone, it has been particularly popular since the 1970’s, no doubt due to its bright colors and relative affordability. Tourmaline gems are found in every color, showing nearly infinite subtle variations in color. Today the majority of Tourmalines are mined in Brazil and various African countries.
Magnetism in Gemstones
An Effective Tool and Method for Gem Identification
© Kirk Feral